Some clinics are already promoting terahertz therapy as safe. But can small pilot studies really support that claim? Have you wondered how solid the evidence is?
Terahertz therapy (uses terahertz waves, a high-frequency, non-ionizing form of electromagnetic energy that doesn’t strip electrons from atoms) usually sounds less risky than treatments that are ionizing. That lowers immediate safety concerns, sure. But lower concern is not the same as proof.
Right now the clinical data is thin. Most studies are single-center pilots with about 10 to 30 people. That’s like a few drops in a big ocean of what we need to know.
A handful of reports show quicker wound healing or better skin repair. The effects can feel like a warm sunrise waking up your cells, but they’re often short-lived and rarely repeated by independent teams.
For the moment, short sessions seem tolerable. We still don’t have strong proof of long-term safety. Larger randomized trials, agreed dosing guidelines, and longer follow-up are needed before terahertz therapy becomes routine care. Would you want this as standard treatment before we have that?
Clinical Evidence For Terahertz Therapy Confirms Safety

Terahertz therapy is still experimental. Terahertz (THz) waves (electromagnetic waves between microwave and infrared frequencies) have drawn interest because they are non-ionizing, which lowers some safety concerns, but clinical proof is thin. Most papers are early pilots, not large confirmatory trials.
-
Study volume: Peer-reviewed work on terahertz therapy is sparse. Most reports are single-center or pilot studies, so the evidence base is small.
-
Sample sizes: Typical studies enroll very few people, often 10 to 30 participants. That makes it hard to draw solid conclusions or apply findings to broader groups.
-
Reported outcomes: A few early reports show faster wound epithelialization and better skin regeneration. For an example, see 太赫茲波療法促進細胞再生. Results tend to be modest, short-term, and rarely replicated by independent teams.
-
Safety and regulatory gaps: Short-term treatments are usually tolerated and may cause only mild, temporary effects. But comprehensive biosafety data are lacking. There are no standardized dose guidelines or agreed treatment protocols, and medical regulatory approvals are generally absent.
So, is terahertz therapy ready for regular clinical use? Not yet. It looks promising for some superficial skin applications, think of it like a gentle, warm buzz on the surface, but current evidence doesn’t give a firm safety or efficacy verdict. Larger randomized trials, standardized dosimetry, and longer follow-up are needed before terahertz moves from experimental to routine care.
Key Clinical Trials of Terahertz Therapy

Clinical trials of terahertz therapy (a type of electromagnetic treatment using terahertz frequencies) have mostly looked at safety and short-term benefits for skin repair and pain relief. They track things like wound epithelialization, lab markers of skin regeneration, and changes in pain scores. Most studies compare an active device to a sham control so researchers can see if effects are real. Curious if it helps wounds or eases sore muscles? That’s what these early trials try to answer.
Study designs vary. Some are randomized and double-blind, others are single-blind or open-label pilot work. Cohorts are small, usually under 30 people, and many studies come from a single center. Device settings and dose – the dosimetry – differ from study to study, so it’s a bit like comparing apples to oranges. Oh, and follow-up windows tend to be short, so long-term effects remain unknown.
| Study Name | Indication | Design | Sample Size | Primary Outcome & Statistical Significance |
|---|---|---|---|---|
| Single-Center Wound Pilot | Wound healing (epithelialization) | Single-blind, sham-controlled | 18 | Faster epithelialization by day 14; reported p < 0.05; no serious adverse events |
| Double-Blind Skin Regeneration Trial | Post-procedural skin regeneration | Double-blind, placebo-controlled | 24 | Moderate improvement in skin texture and collagen markers; reported p ≈ 0.04 |
| Acute Pain Pilot Study | Acute musculoskeletal pain | Randomized, single-blind, sham-controlled | 15 | Reduced pain scores at 1 week; trend toward benefit, p ≈ 0.06; no serious events |
So what do we take away? The trials show modest benefits and a reassuring short-term safety signal. But there are clear limits: samples are small (often 10–30 participants), many studies come from a single site, and endpoints are mixed – that weakens statistical power. Device parameters change across trials, follow-ups are brief, and there are relatively few placebo-controlled studies and even fewer multi-center randomized trials. In truth, these findings are preliminary and need confirmation in larger, well-powered studies before we can say much for sure.
Safety Profile and Adverse Effects of Terahertz Therapy
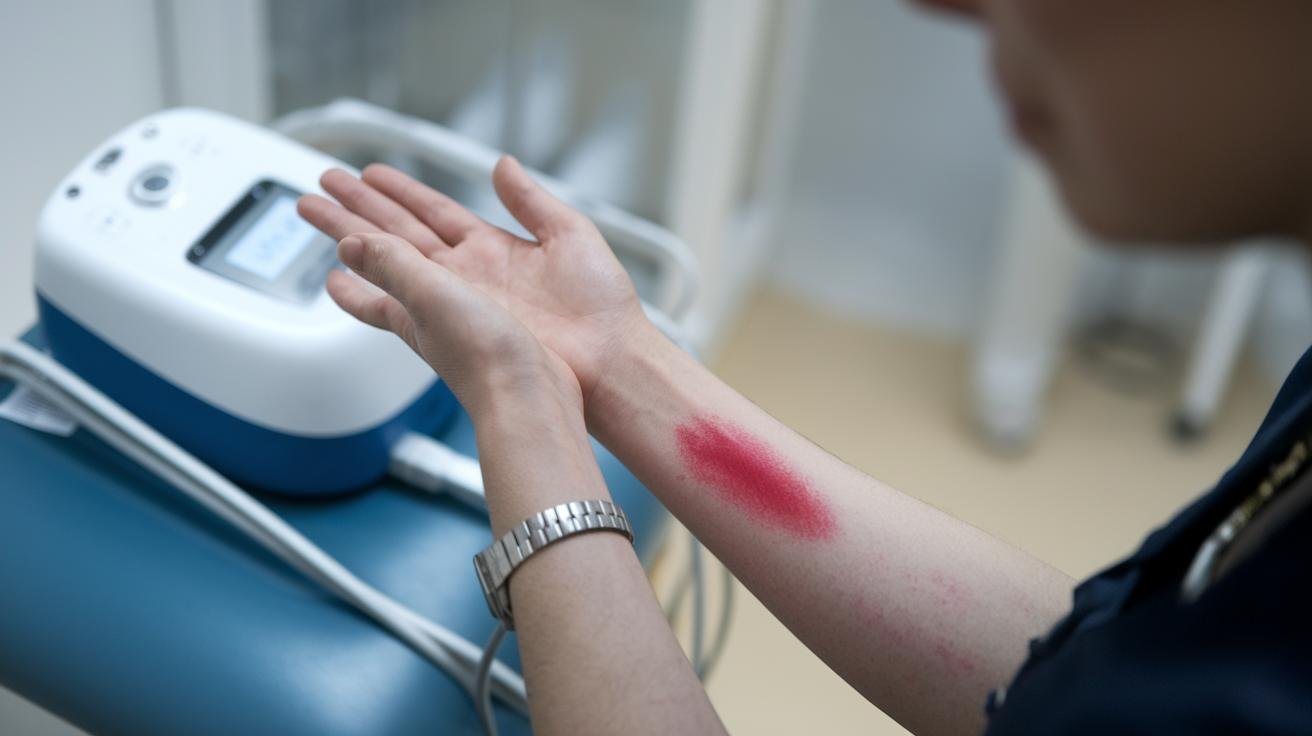
Most reported side effects from terahertz therapy (terahertz = electromagnetic waves sitting between microwaves and infrared light) are mild and short lived. You might notice a light redness or small skin irritation where the device touched you. A gentle warm feeling, like sun on your skin, is common. Some people feel tired for a day or two, usually one to three days.
A few users say they had a temporary bump in heart rate or blood pressure. Eye irritation can happen if you skip protective eyewear. Rare allergic reactions have been reported, such as a mild rash or shortness of breath. Keep in mind most data come from short trials or consumer reports, so the number of cases is small and rare problems might not show up yet. Wait, let me clarify that: limited data means we can’t be certain about every possible rare effect.
Take extra care if you’re pregnant or nursing, have a pacemaker or other electronic implant, have active skin conditions or a photosensitivity disorder, or if you’re a child or teen. These groups should talk with a clinician before trying terahertz therapy.
Current safety guidance leans on standards like IEEE C95.1–2005 and ANSI Z136.1–2007 (model-based exposure limits), not large long-term human trials. That leaves open questions about long-term effects, consistent dosing, and how different devices are tested.
In truth, being cautious makes sense while the evidence grows. Wear eye protection, tell your provider about implants or pregnancy, and watch how your body reacts after a session. If something feels off, stop and check with a healthcare professional. Oh, and a neat little tip: take a quick photo of the treated area before your session so you can spot any changes later.
Treatment Protocols and Dosage Guidelines in Terahertz Therapy
Terahertz therapy uses terahertz waves (electromagnetic waves between 0.1 and 10 THz). These waves are non-ionizing and mostly absorbed near the skin’s surface. Imagine a faint, warm touch on your skin rather than a deep zap. Have you ever felt that gentle heat from a lamp? It’s similar.
Typical sessions people report run 10 to 30 minutes total. Practitioners often make focused passes of about 1 to 5 minutes on each target area. But here’s the catch: researchers don’t yet have a clear dose-response curve for terahertz therapy. That means studies show a wide range of power settings and exposure times. So the numbers you see can vary a lot.
A lot of consumer “terahertz” devices mostly give off infrared heat instead of true THz radiation. So the frequency advertised may not match what the device actually outputs. In truth, don’t assume the label is the same as the measured output.
Device placement and how often you repeat treatment tend to be conservative in existing reports. Keep the emitter about 8 to 10 inches from your skin, and limit use to roughly 2 to 3 sessions per area each day. Start at the lowest comfortable intensity and keep passes short. Watch the skin for redness or sensitivity. Oh, and here’s a neat trick: test a small patch first before treating a larger area.
The mechanisms behind terahertz effects are still being teased apart. Some effects seem thermal, like gentle warming. Others may be subtle, non-thermal interactions with cell structures. Which of those matters clinically at low device power is still unclear. Because standardized dosimetry, reporting, and treatment protocols are missing, treat current schedules and settings as provisional. In short, be cautious and expect guidance to change as larger trials give clearer answers.
Regulatory Landscape for Terahertz Therapy

No terahertz therapy device has FDA approval or clearance for medical use, so claims of medical benefit aren't backed by mandatory regulatory review.
Many terahertz products are sold as wellness tools. That means companies can market them without proving safety or clinical effectiveness. So buyers often end up relying on small lab studies, pilot trials, or company testing instead of a formal regulatory review. It can feel like reading glossy brochures and trusting a few data points.
By contrast, some PEMF (Pulsed Electromagnetic Field therapy) modalities have received FDA clearance for specific uses. For a side-by-side look, see terahertz therapy vs PEMF therapy.
If you’re thinking about trying a device, ask for peer-reviewed studies, independent safety data, and clear instructions for use. A little caution goes a long way.
🎁 Get a Free Wellness Gift
Enter your name and email below to receive a special wellness gift from OlyLife — absolutely free!
Mechanistic Insights into Terahertz Therapy
Terahertz (THz) waves sit between microwaves and infrared on the electromagnetic spectrum, roughly 0.1 to 10 THz. They don’t carry enough energy to ionize tissue. They’re strongly absorbed by water, so most of the energy is taken up in the first millimeter of skin rather than delivered deep into muscle. Think of it as a near-surface effect, like a soft, steady warmth on the skin instead of a deep massage.
Some effects are clearly thermal. You get localized warmth, small rises in tissue temperature, and the microvascular changes you’d expect from gentle heating. It’s a little like a warm sunrise waking up your cells. Relax. Breathe.
At the same time, scientists are exploring possible non-thermal effects , subtle shifts in cell signaling or structural proteins that happen without obvious heat. Examples include changes in inflammatory markers or reorganization of the actin cytoskeleton (the cell’s internal scaffolding). Wait, let me clarify that separating true non-thermal actions from heat-driven ones is tricky, because even slight warming can trigger similar changes.
In lab models, high-power terahertz exposures have produced measurable cellular responses: altered neural stem cell proliferation, transient DNA strand breaks, shifts in gene expression, and disruption of actin networks in some cell types. Those results highlight potential risks if energy is concentrated or used improperly. In other words, under intense conditions, cells can respond in ways we don’t want.
But at common consumer or clinical intensities, the picture is murkier. Most devices run far below the power levels used in those lab studies, and it’s unclear which lab findings matter in living tissue. We need careful, controlled dose-response studies that measure both thermal and non-thermal endpoints, plus longer follow-up in vivo, before we can say which observed lab effects matter for real-world use. Oh, and if you’re trying a terahertz device, look for data showing skin temperature changes and longer-term safety monitoring.
Research Gaps and Future Directions in Terahertz Therapy

Below is a clear, usable research agenda to help THz (terahertz) devices move from small pilots to trustworthy clinical evidence. Think of this as a checklist you can follow when planning the next round of studies.
-
Do systematic reviews and meta-analyses first.
Pool the small pilots to estimate overall effect sizes and check how consistent results are across studies. Think of it like assembling tiles into a mosaic so you can see the bigger picture. Pre-register the review protocol so the methods are transparent. -
Create a CONSORT-style reporting checklist plus a dosimetry standard.
CONSORT (Consolidated Standards of Reporting Trials) gives a clear format for reporting trials, and we need a THz version that includes dosimetry (how much energy is delivered). Report device make and model, waveform, power density, calibration method, exposure geometry, and any firmware or software settings. Treat it like an ingredient list so others can repeat the recipe. -
Run multi-center, pre-registered randomized controlled trials with justified sample sizes and at least 12-month follow-up.
Pre-register the sample size calculation, primary endpoint, and that 12-month follow-up plan. Multi-site studies boost generalizability and make recruitment easier. Power the trials so they can detect realistic effects. -
Require independent calibration, common outcome measures, and open data.
Have devices calibrated by an independent lab and use shared outcomes like wound closure metrics, validated pain scales, and a standard adverse-event taxonomy. Deposit deidentified datasets in a public repository so researchers can pool data later and reanalyze results. -
Set up safety monitoring guidelines and registries for long-term tracking.
Establish clear safety surveillance rules during trials and create national or international device registries to capture late adverse events and device interactions. A registry is the best way to spot rare or delayed problems once devices are in wider use. -
Include diverse populations and cohorts with common contraindications.
Enroll people from different ages, races, and health backgrounds, and run focused cohorts for pregnancy and implanted devices to understand real-world risks. Safety data must reflect the people who’ll actually use these devices.
Next, apply this agenda directly to the trial gaps and limitations noted earlier rather than repeating those study details here. These steps will help move THz research from small, noisy pilots to solid, actionable evidence.
結語
In the action of this post we walked through the current, mostly experimental clinical work on terahertz therapy: small studies, preliminary outcomes, safety notes and missing protocols.
We listed trial sizes, common mild effects, frequency and dosing variability, and gaps in regulation and long-term data.
The clinical evidence for terahertz therapy is limited but encouraging, early signs point to wound and skin repair, mild pain relief and possible recovery benefits. With careful, larger trials we can find clearer answers and better ways to support deep relaxation and recovery.
常問問題
Terahertz therapy side effects
Terahertz (0.1–10 THz) therapy side effects include mild skin irritation, localized warmth, brief fatigue, occasional temporary heart-rate or blood-pressure rises, and rare allergic rashes or breathing difficulty; most resolve within one to three days.
Is terahertz therapy safe
Terahertz (0.1–10 THz) therapy safety is not established; short-term reports show mostly mild, reversible effects, but long-term safety data and standardized dosing are lacking, so caution is advised for pregnant people, children, and pacemaker users.
Terahertz therapy for stroke patients
Terahertz (0.1–10 THz) therapy for stroke patients has very limited study; small pilot trials are sparse and show no clear, replicated benefit, so it remains experimental and shouldn’t replace standard stroke care.
Terahertz Therapy Machine
A terahertz (0.1–10 THz) therapy machine is a device claiming to emit THz energy; many consumer “blower” models mainly produce infrared heat, user reviews vary, and clinical validation plus regulatory clearance are generally absent.
Is terahertz therapy real
Terahertz (0.1–10 THz) therapy is real as an experimental modality; lab and small clinical studies report biological effects, but robust, large-scale trials and regulatory approvals are missing, so clinical use is limited.